“The application of edaravone dexborneol can significantly reduce the level of inflammatory factors and improve the neurological function in patients with AIS, either received or not received alteplase thrombolytic therapy.”
The conclusion was drawn from a retrospective clinical study published in a poster at the 2022 European Stroke Organization Conference (ESOC) held from May 4 th to 6th in 2022.
Led by the corresponding author Dr. Lixin Xu, the clinical team from Department of Neurology, Dezhou People’s Hospital in ShanDong China analyzed the nerve function recovery and changes of inflammatory factors in patients with acute ischemic stroke (AIS) , who have received four different treatment regimens.

Title:
Effects of Edaravone Dexborneol on Neurological Function and Serum Inflammatory Factors in Patients with Acute Ischemic Stroke
Abstract number:
P0009
Nearly 200 cases of AIS treated from September 2020 to April 2021 with similar baseline characteristics were analyzed retrospectively. Patients were divided into 4 groups: edaravone dexborneol group (n = 65), routine treatment group (n = 68), edaravone dexborneol combined with alteplase thrombolytic group (n = 29) , and alteplase thrombolytic group (n = 35).
The changes of National Institutes of Health Stroke Scale (NIHSS) score, high-sensitivity C-reactive protein (hs-CRP), interleukin-6 (IL-6) and tumor necrosis factor (TNF- α) before and after treatment were compared between groups.
At day 90, the changes before and after treatment of NIHSS score, hs-CRP, TNF-α, and IL-6 levels were significantly different between four groups. After a pairwise comparison, the NIHSS score, hs-CRP, TNF-a, and IL-6 in the combination treatment group decreased more than other three groups (P<0.05). Patients in both the edaravone dexborneol group and alteplase thrombolytic group had higher decrease in both NIHSS score, hs-CRP, TNF- a, and IL-6 than the routine treatment group(P<0.05).
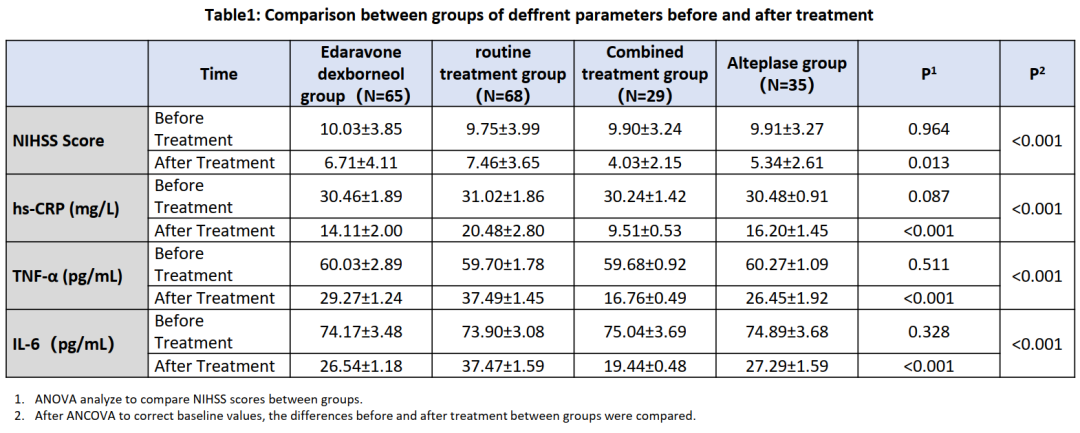
The result implies that the application of edaravone dexborneol can significantly reduce the level of inflammatory factors and improve the neurological function in patients with AIS, either received or not received alteplase thrombolytic therapy. This work supported the clinical application of edaravone dexborneol for AIS treatment.
Sanbexin® (edaravone dexborneol)is the only innovative drug for stroke approved worldwide since 2015. The studies with clinical evidence of Sanbexin® has been included in ESOC for two consecutive years and allow the international academic community to further understand this new product independently developed in China.