March 30, 2026 — Simcere Pharmaceutical Group Limited (2096.HK) announced that Phase III results from the RADIANT-AD study of rademikibart, an investigational monoclonal antibody developed in collaboration with Connect Biopharma, were presented as a Late-Breaking Abstract (LBA) oral presentation at the 84th Annual Meeting of the American Academy of Dermatology (AAD) in Denver, United States.
The study, led by co-principal investigators Professor Zhang Jianzhong and Professor Zhou Cheng of Peking University People’s Hospital, evaluated rademikibart in adults and adolescents with moderate-to-severe atopic dermatitis (AD). Results showed that rademikibart provided rapid relief of itch and improvement in skin inflammation within two weeks, along with high levels of skin clearance that were sustained through 52 weeks. The findings were presented by Professor Zhou Cheng on behalf of the research team on March 28.
Dr Zhou Cheng giving oral presnetation at the 84th AAD Annual Meeting
Atopic dermatitis is a chronic, relapsing inflammatory skin disease characterized by recurrent eczematous lesions, severe dryness, and intense pruritus. It is estimated that approximately 70 million people in China are affected, with nearly half experiencing moderate-to-severe disease. Despite the availability of biologic therapies, disease management remains challenging, and there is a continued need for more effective and durable treatment options.
The randomized, double-blind, placebo-controlled Phase III trial (NCT06477835) enrolled 259 adult and adolescent patients with moderate-to-severe AD. Participants were randomized in a 1:1 ratio to receive rademikibart or placebo for 16 weeks, followed by a 36-week open-label extension during which all patients received rademikibart.
Efficacy Results
At Week 16, a significantly greater proportion of patients treated with rademikibart achieved key endpoints compared with placebo (p<0.0001), including:
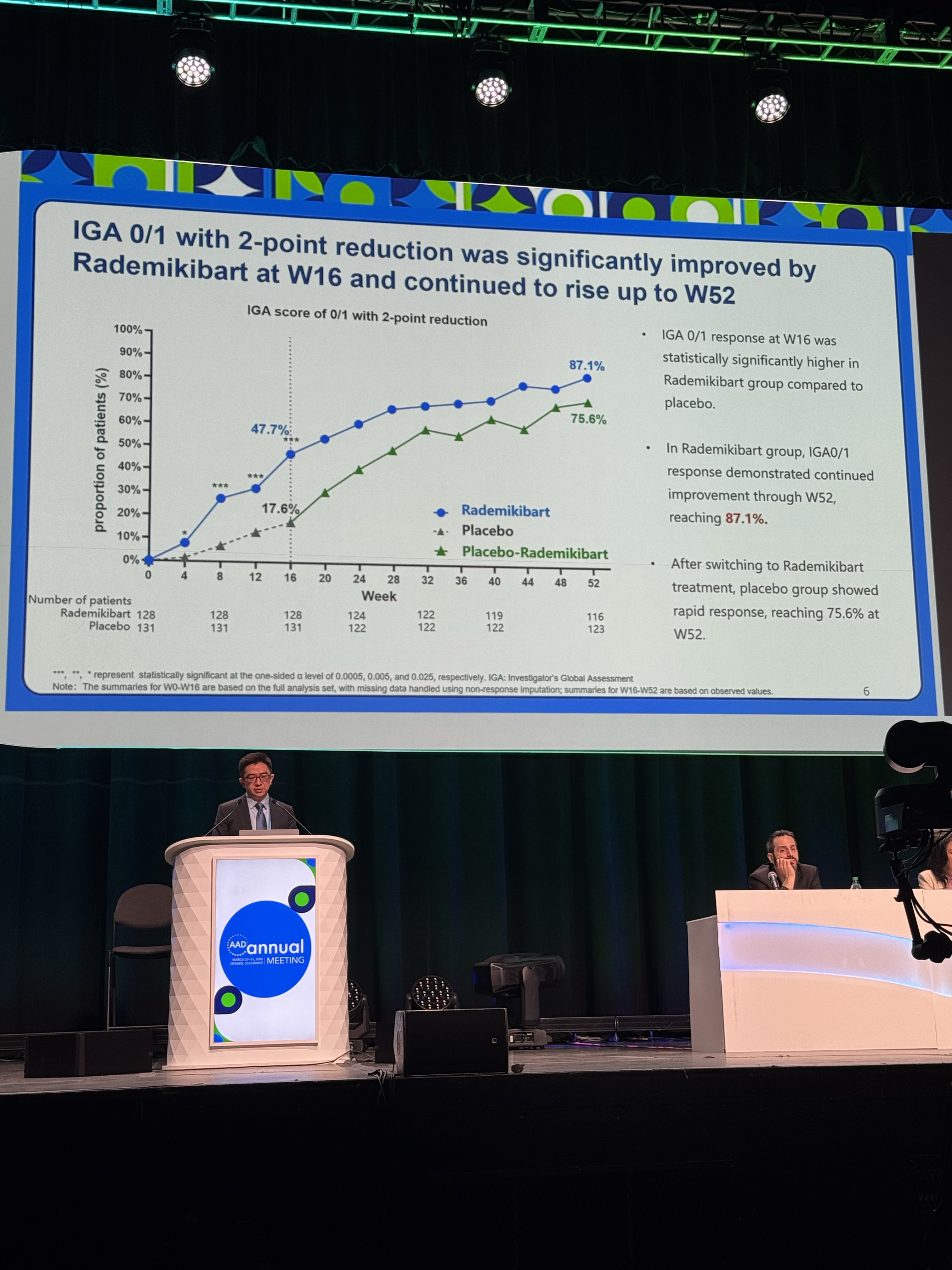
Investigator’s Global Assessment (IGA) score of 0 or 1 with a ≥2-point reduction from baseline (47.7% vs. 17.6%)
≥75% improvement in Eczema Area and Severity Index (EASI-75) (74.2% vs. 34.4%)
≥90% improvement in Eczema Area and Severity Index (EASI-90) (43.0% vs. 14.5%)
≥3-point reduction in Peak Pruritus Numerical Rating Scale (PP-NRS) (54.7% vs. 27.5%)
Efficacy was sustained and further improved through Week 52, with response rates reaching 87.1%, 96.6%, 85.3%, and 91.2% for the respective endpoints.
Safety
Rademikibart demonstrated a favorable safety profile over 52 weeks, consistent with other anti–IL-4Rα therapies.
At Week 16, the incidence of treatment-emergent adverse events (TEAEs) was 60.9% in the rademikibart group and 64.9% in the placebo group. By Week 52, the overall incidence of TEAEs was 82.2%. Rates of serious adverse events (SAEs) and treatment discontinuations due to adverse events remained low throughout the study period.
Professor Zhang Jianzhong commented:
“Rademikibart demonstrates the dual advantages of strong efficacy and a favorable safety profile, with its long-term safety further strengthening clinical confidence. From a clinical perspective, its significance extends beyond providing a new treatment option; it has the potential to help raise treatment goals for moderate-to-severe atopic dermatitis and support standardized, long-term disease management.”
Professor Zhou Cheng added:
“Rademikibart showed the ability to deliver deep and sustained disease control in atopic dermatitis, with rapid onset and continuously improving efficacy over one year of treatment, without an apparent plateau. These findings address key challenges in long-term disease management and suggest that rademikibart may offer an effective and well-tolerated biologic option for patients.”
Rademikibart is a fully human monoclonal antibody targeting IL-4Rα. In preclinical studies, it has demonstrated high binding affinity and inhibits the signaling of both IL-4 and IL-13, key drivers of type 2 inflammation. By targeting this pathway, rademikibart is being developed for the treatment of inflammatory diseases including atopic dermatitis and asthma.
In November 2023, Simcere entered into an exclusive licensing and collaboration agreement with Connect Biopharma, obtaining rights to develop, manufacture, and commercialize rademikibart for all indications in China. The Phase III study was conducted across 59 clinical centers in China and successfully met its primary endpoint.
The Phase III results provide supportive evidence of rademikibart’s clinical profile. In July 2025, the New Drug Application (NDA) for rademikibart was accepted for review by China’s National Medical Products Administration (NMPA), potentially expanding treatment options for patients with atopic dermatitis in China.